 Our work on understanding the energy flow in complex molecules has been published in Journal of Molecular Spectroscopy. We provide an efficient and reliable method for modelling microwave spectra that have been difficult to analyse and extract reliable physical information from, especially for complex molecules such as toluene which contains a rotating methyl group within its structure. The software developed to predict rotational energy changes in complex molecules such as toluene is available for use by other researchers working on rotational spectroscopy problems. Please contact Dr Jason Gascooke for more information.
Our work on understanding the energy flow in complex molecules has been published in Journal of Molecular Spectroscopy. We provide an efficient and reliable method for modelling microwave spectra that have been difficult to analyse and extract reliable physical information from, especially for complex molecules such as toluene which contains a rotating methyl group within its structure. The software developed to predict rotational energy changes in complex molecules such as toluene is available for use by other researchers working on rotational spectroscopy problems. Please contact Dr Jason Gascooke for more information.
- “The effects of torsion–vibration coupling on rotational spectra: Toluene reinterpreted and refitted”, J.R. Gascooke and W.D. Lawrance, J. Molec. Spectrosc. 318, 53 (2015). doi:10.1016/j.jms.2015.10.006 http://www.sciencedirect.com/science/article/pii/S0022285215300060
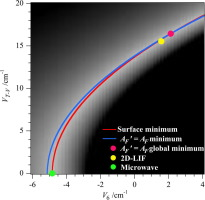
We have taken high-precision microwave absorption data for toluene from the literature and reanalysed it using a model that incorporates coupling between the methyl (CH3) torsion motion and the lowest energy vibration. From our previous analysis of 2D-LIF electronic spectra of toluene we find that we require such a coupling, however, the precise microwave data (due to changes in overall rotation of the molecule) was fit without it. We demonstrate that the microwave data can be fit equally as well by either including or excluding the coupling, and show why this is possible. Importantly, we find the barrier height to decrease substantially when the coupling is included, changing from 4.9 cm-1 to 1.8 cm-1 and the minimum energy conformation changes from the methyl hydrogens being staggered (relative to the phenyl plane) to eclipsed.